OUR PROGRAMS
-
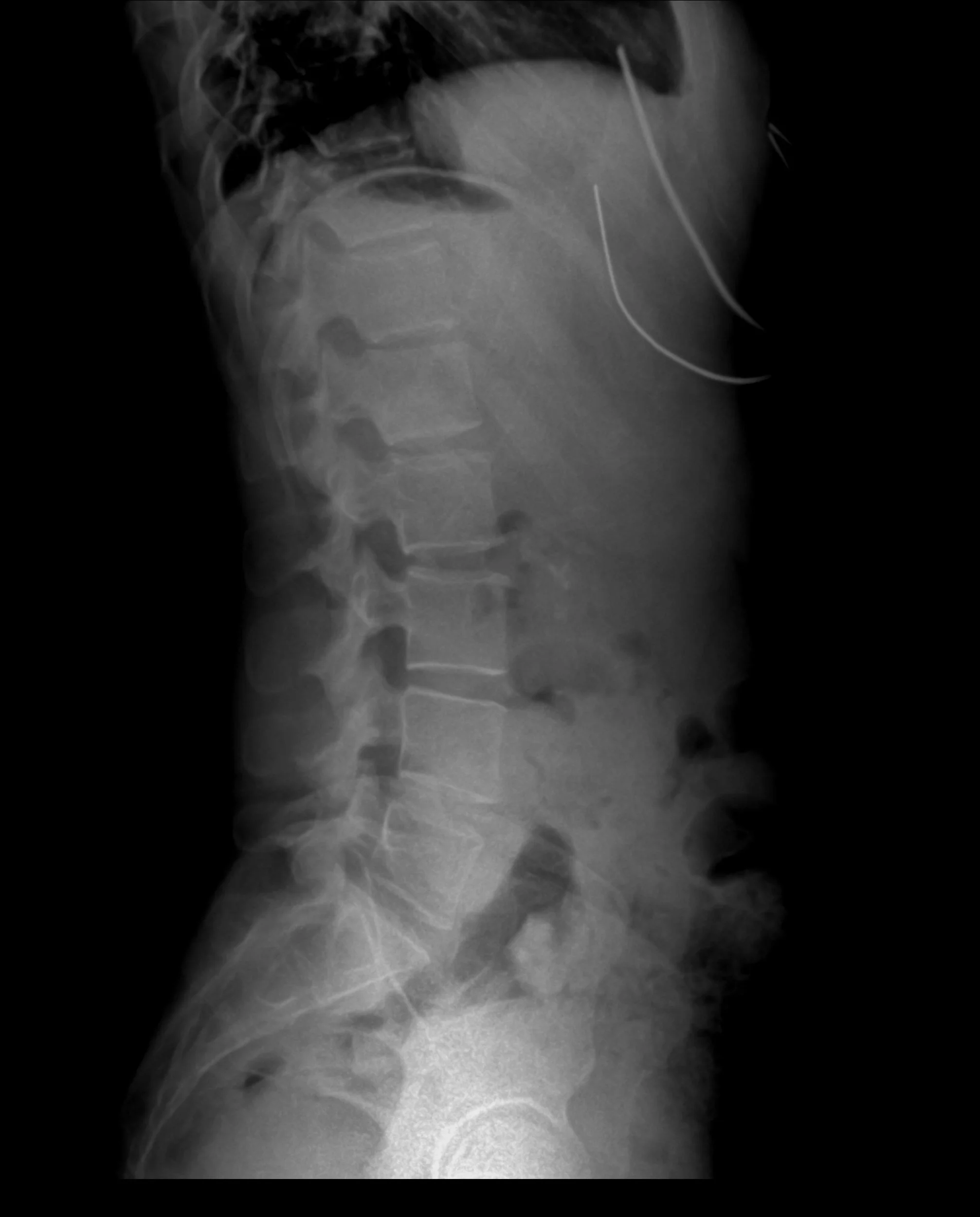
CellTop
This study is designed to assess the safety and feasibility of injecting mesenchymal stem cells (MSCs) derived from a patient’s own fat tissue into the cerebrospinal fluid after a severe traumatic spinal cord injury. It uses autologous, culture-expanded adipose-derived MSCs (AD-MSCs) delivered intrathecally and remains experimental, with the goal of exploring its potential to support recovery in individuals with spinal cord injury.
-
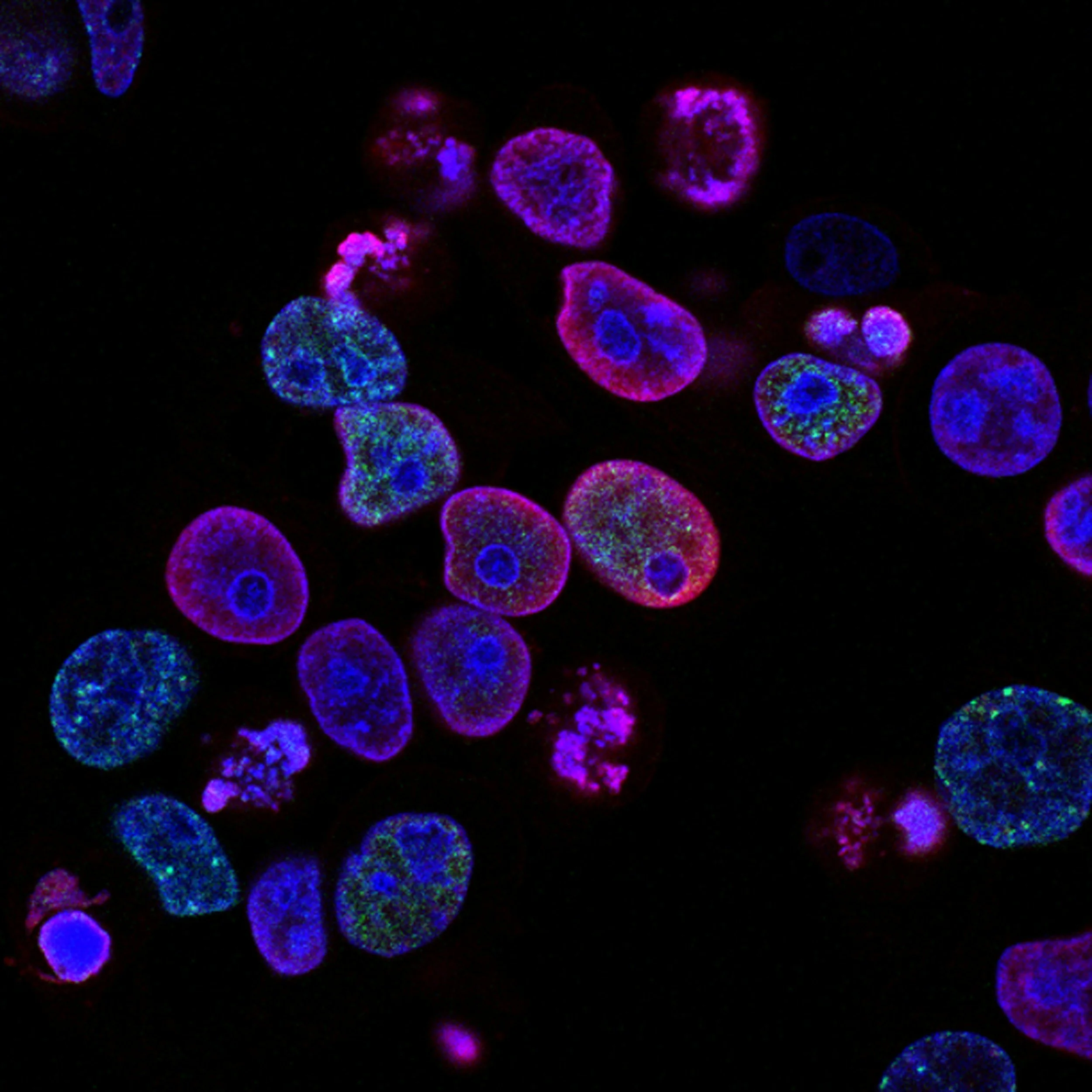
NeuroRise
This study evaluates the safety and tolerability of intrathecal allogeneic umbilical cord-derived mesenchymal stem cells (UC-MSCs) in patients with traumatic spinal cord injury. It will also collect functional recovery outcomes after administration to generate preliminary evidence to inform a future Phase II efficacy trial.
